Volume 10 - Year 2023 - Pages 20-23
DOI: 10.11159/jbeb.2023.004
Suggestive Decreasing Effects of the COVID-19 Pandemic on Reported Adverse Arrhythmic Events and 30-Day Fills For Anti-Arrhythmic Agents
Eshaan Gandhi1, Sujata Bhatia2
1Indian Hill High School
1 Beaufort Hunt Lane, Cincinnati, United States of America
eshaan.gandhi24@ihsd.us; sbhatia@g.harvard.edu
2Faculty of Arts and Sciences, Harvard University
51 Brattle St, Cambridge MA 02138, United States of America
Abstract - COVID-19 had large impacts on the lives of many individuals with rhythmical cardiac problems. With limitations that COVID-19 had on the ability to track medical based data, a controversy on the effect of COVID-19 on the incidence of arrhythmic activity has been apparent. To determine the effect that pandemic had on the incidence arrhythmic activity, we studied adverse event trends of 4 anti-arrhythmic agents — propafenone, sotalol, amiodarone, and dronedarone. Extracting data from the FDA FAERS database, we concluded significant (p<0.05) decreases for propafenone (55.8% decrease) and amiodarone and dronedarone (16.9% decrease) from 2020 to 2021 as well as an insignificant decrease for sotalol (30% decrease). In response to suggestive decreasing trends, we proceeded with a cost-analysis to explore possible reasons behind sudden decreases in reported adverse events. Using the Medicare Part D database, data for costs between generic vs. brand-name for previously examined antiarrhythmic agents as well as associations between 30-day fills and adverse event reports was examined. For each of the agents, the brand-name agents had a significantly higher cost than the brand-name agents. Associations between adverse events and 30-day fills were demonstrated through R2 values, which resulted in values of 0.238 for propafenone, 0.796 for sotalol, and 0.651 for amiodarone and dronedarone. We concluded that cost did not have a significant effect on adverse event reporting. Overall, we recommend further study into the potential underdiagnosis and undertreatment of cardiac arrhythmias during the COVID-19 pandemic, which can have dangerous implications on the long-term mortality of individuals without the appropriate materials. To accomplish this, it is necessary to analyze 2022 adverse event data for various anti-arrhythmic agents to observe new or continuing trends.
Keywords: COVID-19, arrhythmia, pharmaceuticals, cost-analysis, adverse events, FDA
© Copyright 2023 Authors This is an Open Access article published under the Creative Commons Attribution License terms. Unrestricted use, distribution, and reproduction in any medium are permitted, provided the original work is properly cited.
Date Received: 2023-08-10
Date Revised: 2023-09-20
Date Accepted: 2023-10-04
Date Published: 2023-10-12
1. Introduction
The COVID-19 pandemic has placed stress on healthcare systems throughout the world. Such stressors included limitations on facilities, a lack of medical personnel, and public health initiatives that were designed to limit the spread of infectious diseases. Along with these limitations, the COVID-19 pandemic has also represented the challenge of balancing chronic disease detection and treatment with infectious disease management, even when the fields cross paths with each other. Similar to the healthcare field, in society, the COVID-19 pandemic placed immense stress on individuals worldwide due to factors like job losses, social isolation, and the loss of loved ones. Such high stress during an overarching period of time can lead to negative health effects, some of which can lead to cardiac dysfunctionalities through arrhythmic activity.
2. Related Work
It has already been established that COVID-19 can have cardiac effects when spreading throughout the body. This means that the COVID-19 infection itself could predispose to cardiac arrhythmias in individuals, or even worsen existing cardiac arrhythmias. However, with many research and hospital facilities unoccupied during the pandemic, there was an inability to track medical-based data. This limitation has led to a current controversy over whether the pandemic led to an increase or decrease in arrhythmic events. On one hand of the argument, it was found that during the pandemic there was a 32% reduction in ventricular arrhythmias needing device therapies, which coincided with measures of social isolation [1]. On the other hand of the argument, it was found that during the pandemic there was an increase in arrhythmic events in major cities. Specifically, when comparing analysis time periods coinciding with the pandemic in 2020 with the same periods in 2019, researchers observed a larger mean rate of defibrillator shock episodes per 1000 patients during the pandemic in New York City (17.8 versus 11.7, respectively), New Orleans (26.4 versus 13.5, respectively), and Boston (30.9 versus 20.6, respectively) [2]. Based on these studies, the pandemic may have had different impacts depending on where an individual lives, such as distinctions between major metropolitan versus rural areas. Another existing argument is that it may have been possible that the COVID-19 pandemic led to underdetection, underdiagnosis, and undertreatment of cardiovascular disease due to a major decline in in-person visits to physicians with strict isolation policies present [3]. With these arguments in mind, the main objective of this study is to determine the impact the COVID-19 pandemic has had on the incidence of cardiac arrhythmias through the examination of reported adverse events for anti-arrhythmic agents, costs of anti-arrhythmic agents, and prescriptions of anti-arrhythmic agents.
3. Methods
Throughout our examination, we extracted data from the Food and Drug Administration (FDA) which maintains the FDA Adverse Event Reporting System (FAERS) web based tool, a database of all reported adverse events for therapeutic biological products and drugs [4]. Drug manufacturers are required to submit adverse event reports to the FDA. Similarly exists voluntary reporters which includes consumers and healthcare professionals. FAERS contains data from both mandatory and voluntary reports. The database classifies adverse events according to drug class and reaction type. In our study, the FAERS database was searched for all reported adverse events from January 1, 2020 through December 31, 2021 for the following four anti-arrhythmic agents: propafenone, sotalol, amiodarone, and dronedarone. Due to the dynamic of the COVID-19 virus and its variants, we also decided to explore the incidence of arrhythmias over the course of 2020, specifically comparing the number of adverse events in the first half of 2020 (January - June) vs. the number of adverse of events in the second half of 2020 (July - December) for each anti-arrhythmic. Adverse events from both brand-name and generic versions of the anti-arrhythmic agents are included in the analysis. For the primary analysis of adverse events, statistical comparisons were completed via a student t-test, with a cutoff for significance at p<0.05. In a cost analysis, we examined data through the Medicare Part D prescriber database which lists the number of 30-day-fills and total cost for each prescription medication on an annual basis [5]. The cost per 30-day fill for each drug was calculated by dividing the total cost of the drug by the number of 30-day fills. By analyzing cost, we were able to gain additional insight on reasons behind trends in adverse event data.
4. Results
4. 1. Adverse Event Analysis for Anti-arrhythmic Agents
This study quantified monthly adverse event reports in 2020 and 2021 for 4 major anti-arrhythmic agents: propafenone (class IC), sotalol (class II), amiodarone (class III), and dronedarone (class III). For all 4 anti-arrhythmic agents, the overall number of reported adverse events decreased from the first half of 2020 to the second half of 2020, and the overall number of reported adverse events decreased from 2020 to 2021. More specifically, propafenone showed a statistically significant 55.8% decrease (p<0.002) in reported adverse events from 2020 to 2021 (Figure 1), as well as a statistically significant 53.1% decrease (p<0.05) in reported adverse events from the first half of 2020 to the second half of 2020. Sotalol exhibited a 30% decrease in reported adverse events from 20201 to 2021 (Figure 2), as well as statistically significant 38.8% decrease (p<0.007) in reported adverse events from the first half of 2020 to the second half of 2020. Amiodarone and dronedarone exhibited a statistically significant 16.9% decrease (p<0.02) in total adverse events from 2020 to 2021 (Figure 3). As suggested by these results, arrhythmias may have been under-treated or under-diagnosed during the COVID-19 pandemic due to underlying factors. Once again, these results may suggest that cardiac arrhythmias have been under-treated or under-diagnosed during the COVID-19 pandemic, only the changes in treatment may have occurred earlier than once believed.
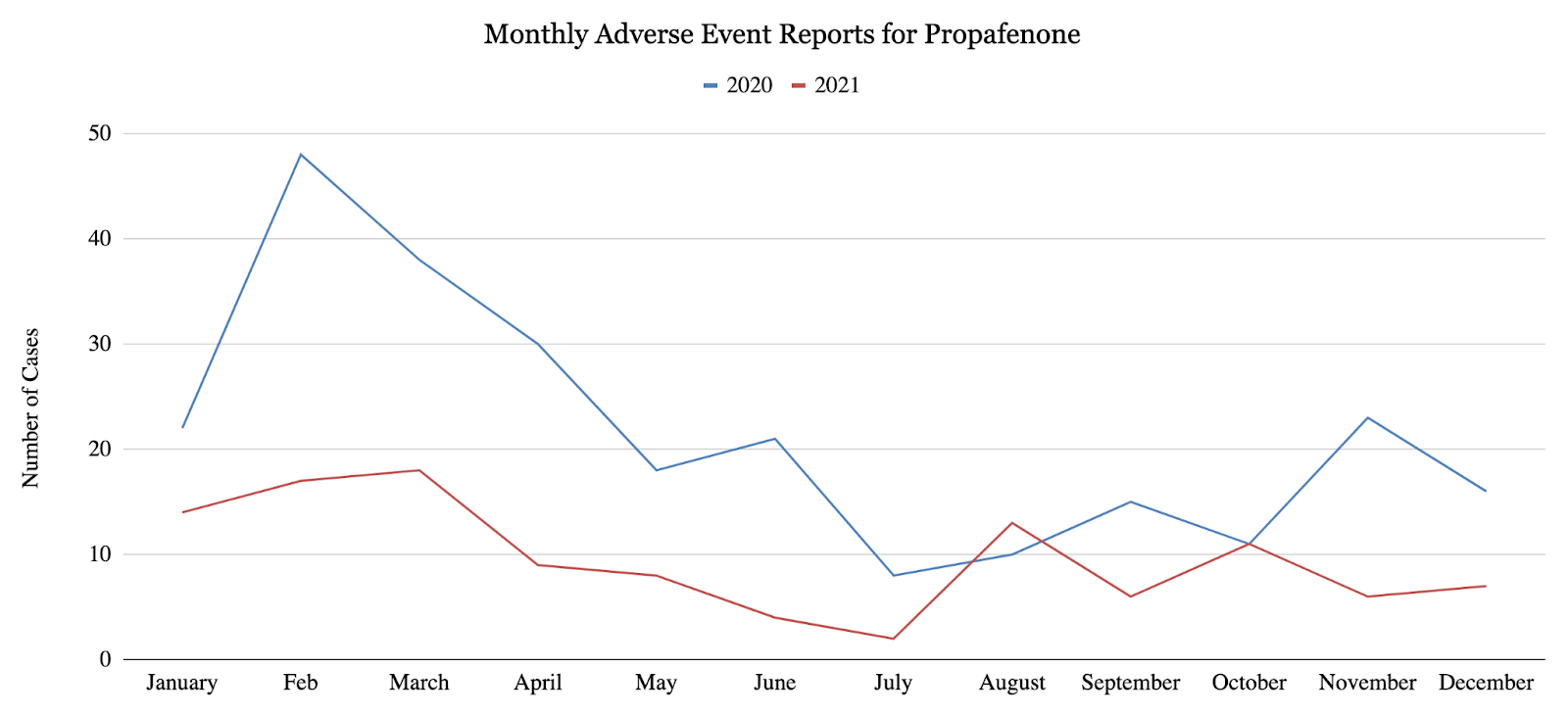
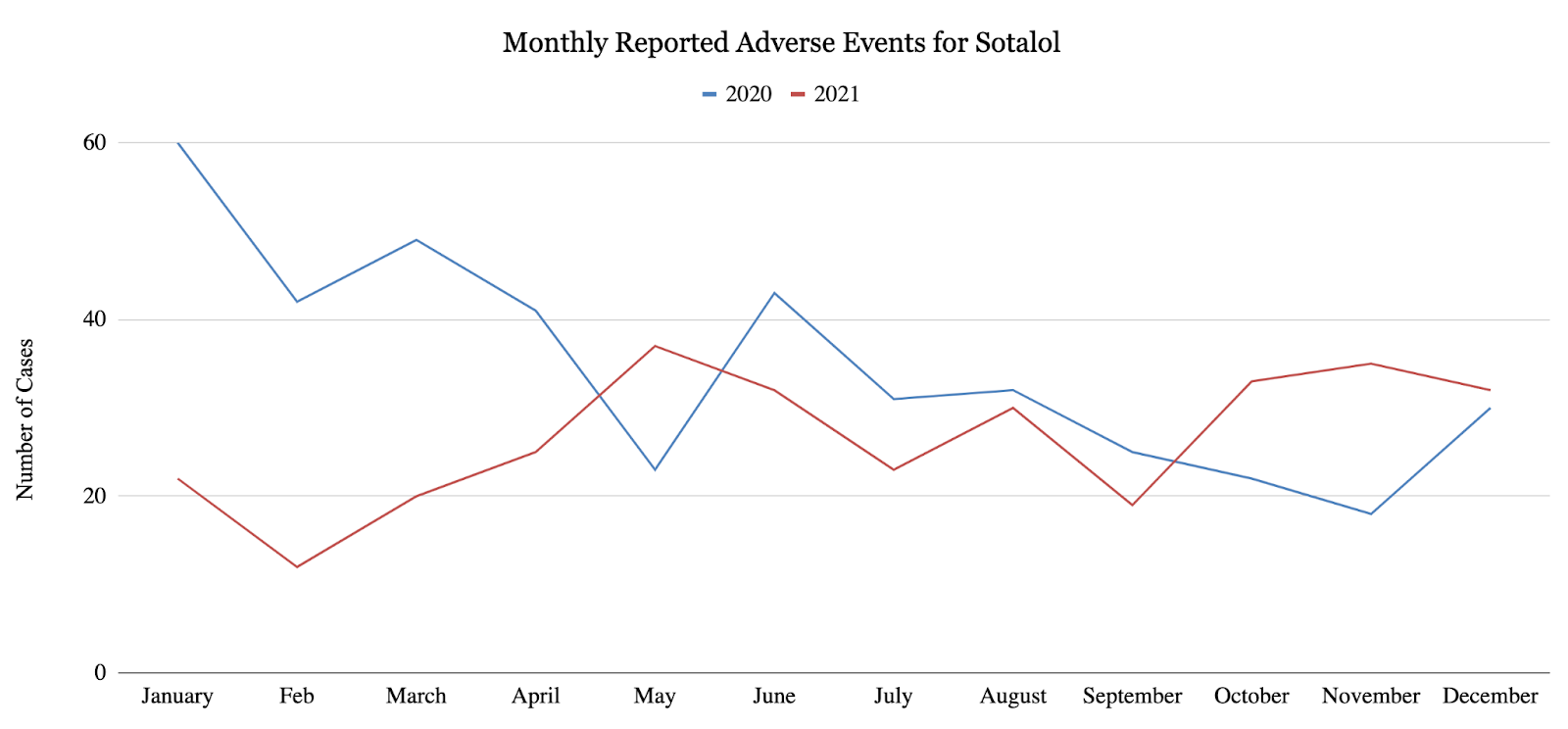
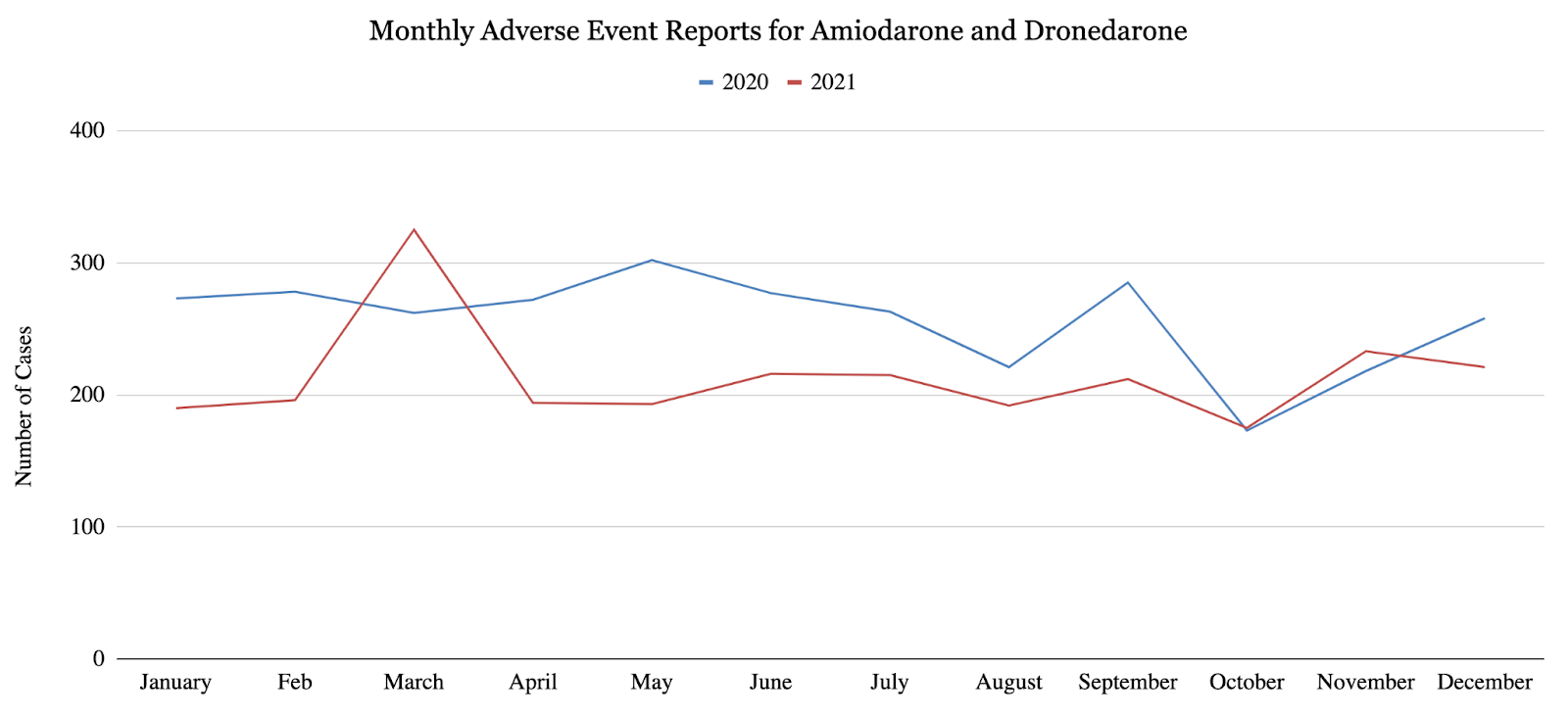
4. 2. Cost Analysis for Anti-arrhythmic Agents
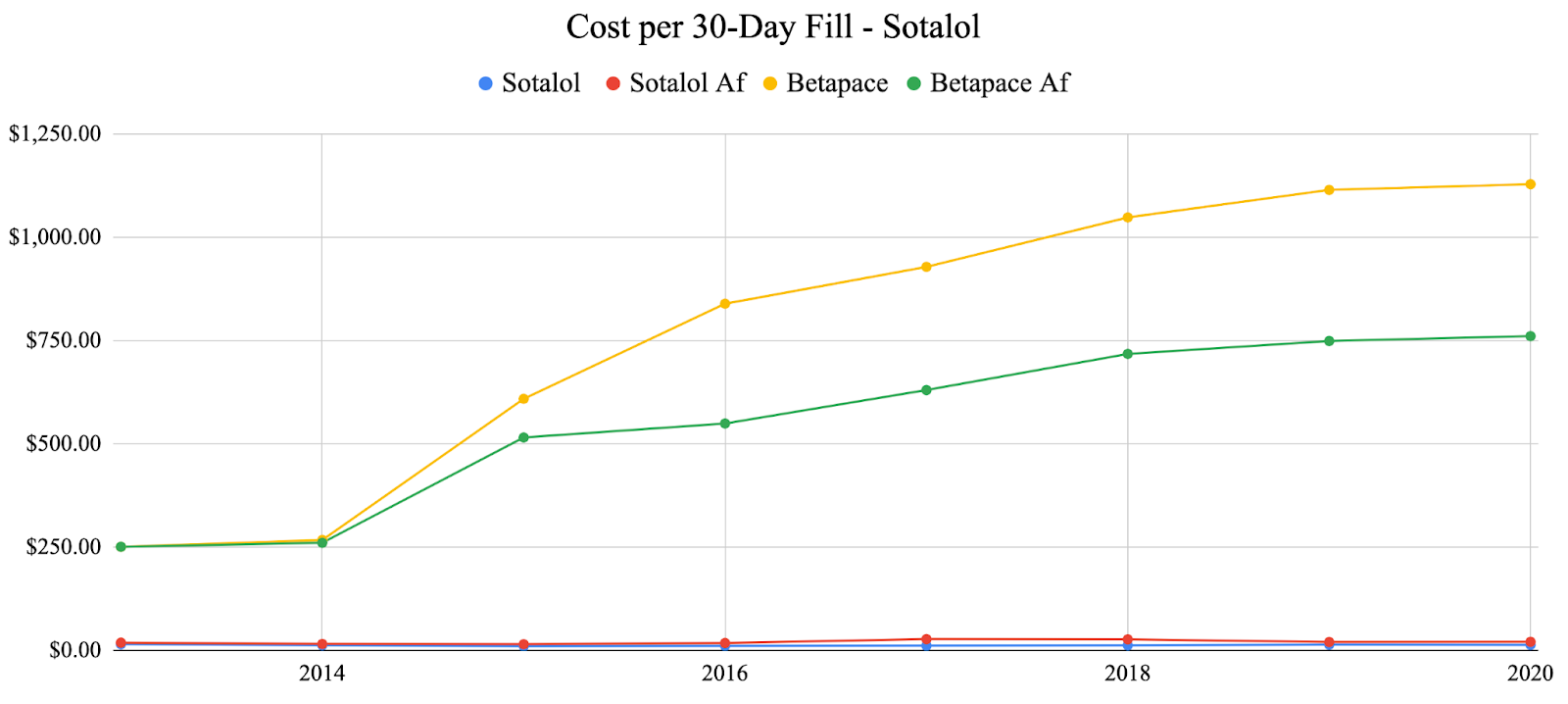
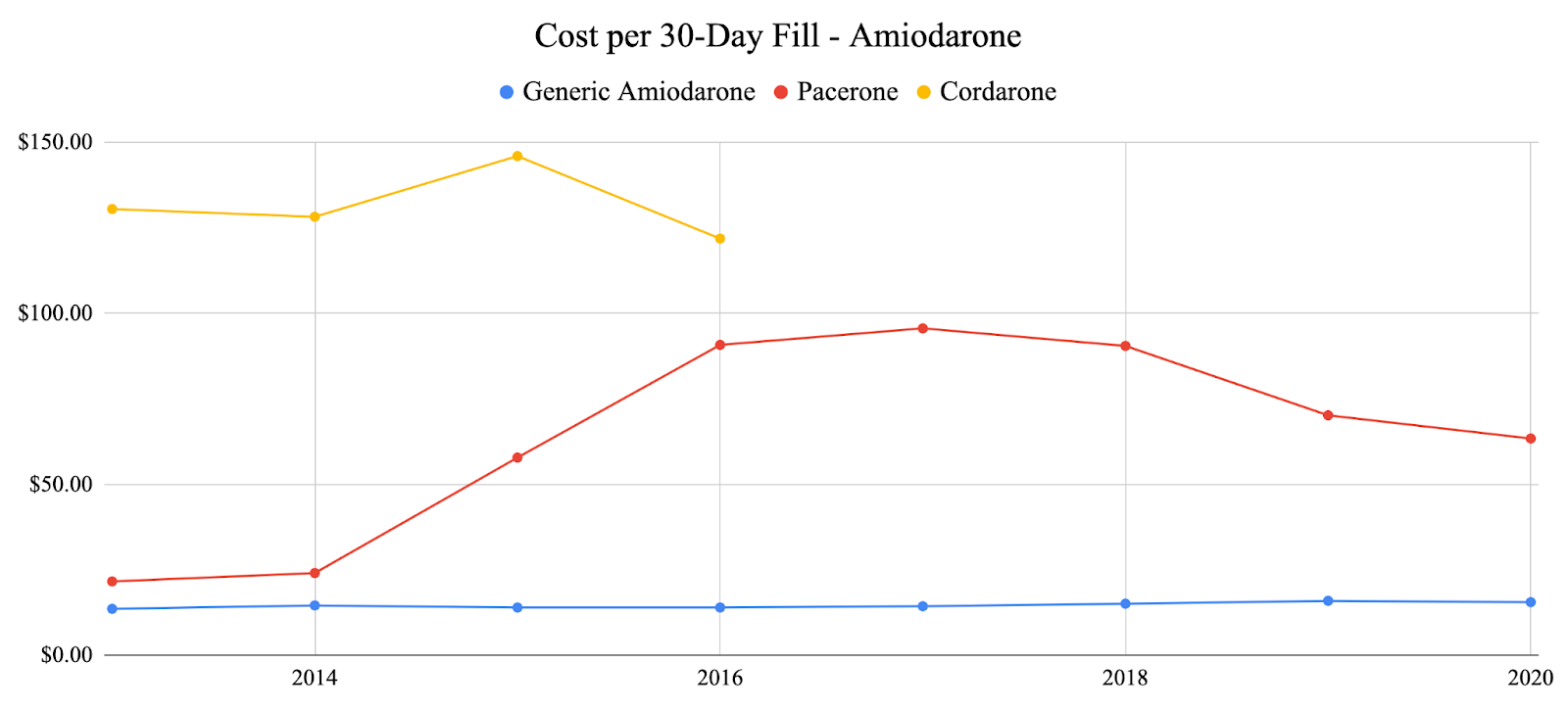
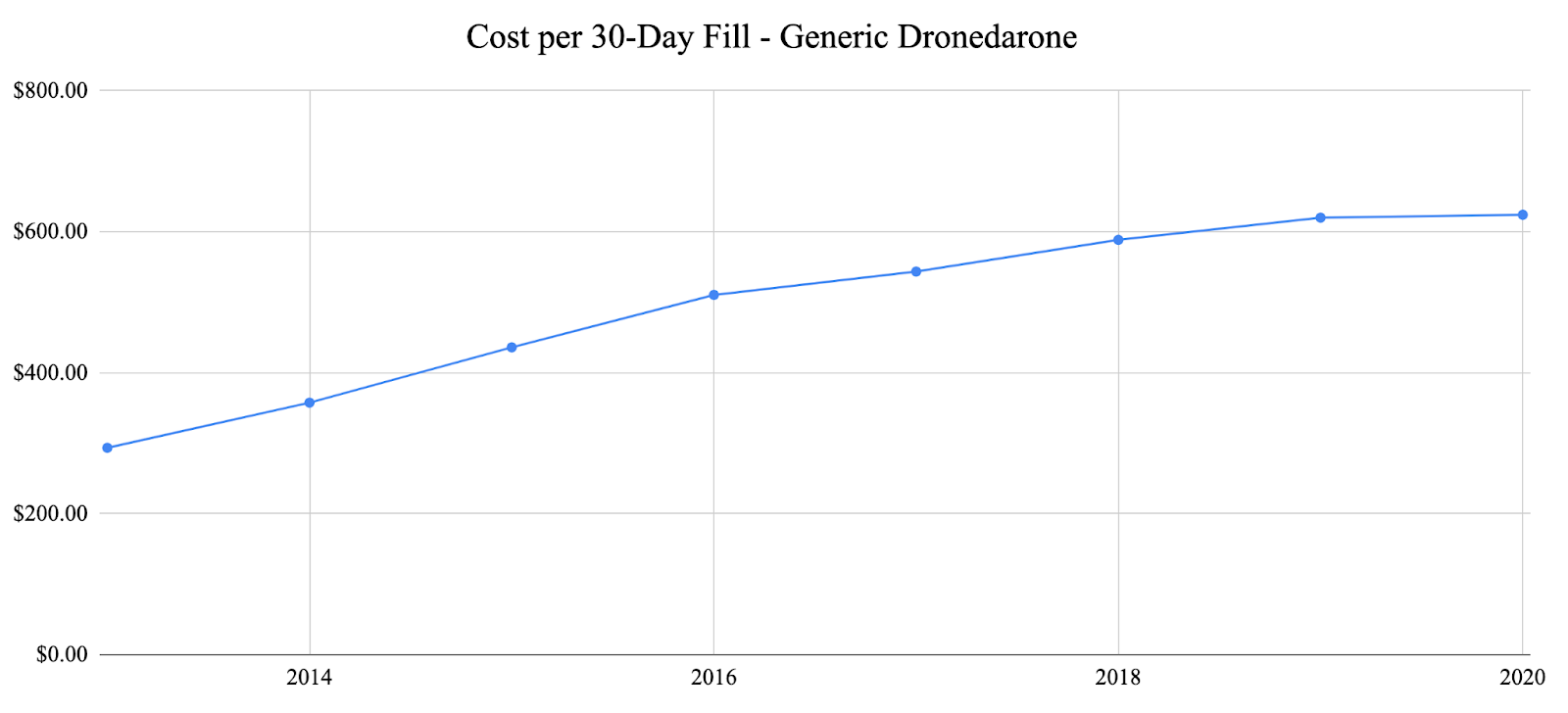
In response to suggestive trends represented by the primary adverse event analysis, we decided to examine one factor that was a prominent issue throughout the COVID-19 pandemic: cost. We specifically examined the costs of previously analyzed generic anti-arrhythmic agents vs. their brand-name counterparts as well as analyzing the total number of 30-day fills vs. the number of adverse events over time for each anti-arrhythmic agent. For each of the agents analyzed, the generic agents were significantly less expensive than the brand-name agents. Generic propafenone has shown variable costs each year, while brand-name propafenone (Rythmol) has shown a steady increase in cost until 2020 where it slightly decreased during the pandemic (Fig. 4). Generic sotalol has shown variable costs each year, while brand-named sotalol (Betapace) has shown continuous growth each year and through the pandemic (Fig 5.) Generic amiodarone has held relatively constant cost each year, while brand-name amiodarone (Pacerone and Cordarone) has shown variable prices, decreasing trends in price starting in 2018 (Fig. 6). Generic dronedarone has shown consistent increasing trends, while no data for brand-name dronedarone was available (Fig. 7). Based on these results alone, it is suggested that cost for generic and brand-name anti-arrhythmic agents were not significantly affected by the pandemic. To investigate further, we analyzed associations between total 30-day fills and total adverse events for each anti-arrhythmic. Amiodarone and dronedarone showed a positive, moderate association with an R2 value of 0.651, propafenone showed a negative, weak association with an R2 value of 0.238, and sotalol showed a positive, moderately strong association with an R2 value of 0.796. Overall, no common trend between total 30-day fills and total adverse events between the studied agents, although filling patterns may be different for each individual agent based on previous supply and demand.
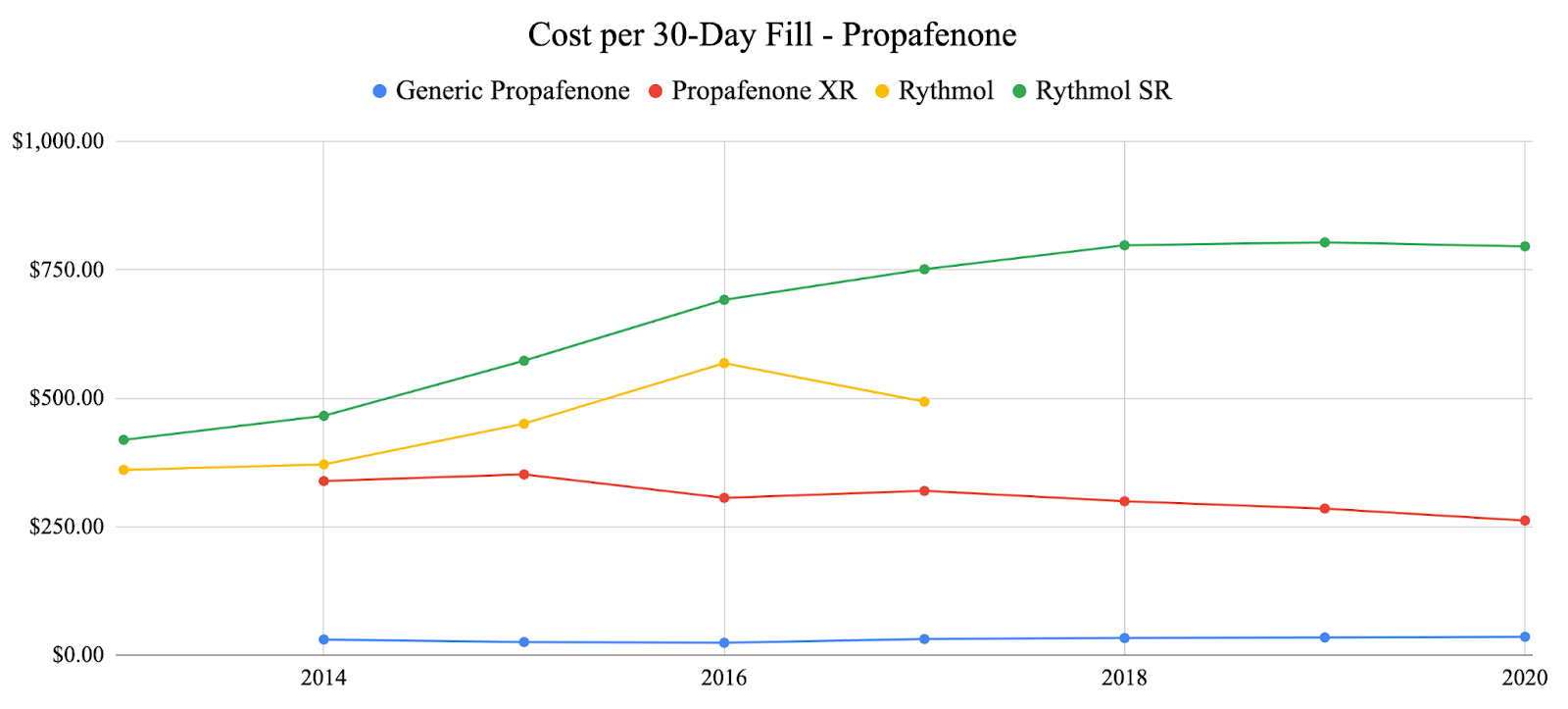
5. Conclusion
Although the adverse event analysis for anti-arrhythmic agents resulted in suggestive trends, there are some limitations that existed that may have manipulated data collected. To start, the number of adverse events reported in the FAERS database may not have reflected the true rate of adverse events. This may have been due to the under-reporting of adverse events during the COVID-19 pandemic with many healthcare-related institutions being inactive. Similarly, there is always the potential for submission of incomplete, inaccurate, or unverified adverse event reports such as the misreporting of adverse events or even duplicate reporting of adverse events. Lastly, unfortunately the FAERS database does not represent all known information or cases for a certain drug product. Irregularities within data exist in many forms, it is the job of an analysis to account for these irregularities and form trends based on the general data. Limitations within cost analysis exist as well. Medicare Part D data only reflects the aggregate data for Medicare Part D recipients, which may signify unrepresented data.
Overall, we observed suggestive decreasing results in adverse event reports for anti-arrhythmic agents before and during the COVID-19 pandemic based on data extracted from the FDA FAERS database. We also observed variable results in cost reports for the respective anti-arrhythmic agents over time which suggested that significant increases in the cost of drugs was not a factor in results for reported adverse events, regardless of the domain of the agent (generic vs brand-name), based on data extracted from the Medicare Part D database. We recommend further study into the potential under-diagnosis and under-treatment of cardiac arrhythmias during the COVID-19 pandemic, which can have dangerous implications on the long-term mortality of individuals without the appropriate materials. To accomplish this, it is necessary to analyze 2022 adverse event data for various anti-arrhythmic agents to observe new or continuing trends.
References
[1] C. J. O'Shea, M. E. Middeldorp, K. Campbell, P. Sanders. Cardiac defibrillator therapies during the COVID-19 pandemic: how you look provides perspective, European Heart Journal, 2022. View Article
[2] S. Adabag, P. Zimmerman, A. Black, M. Madjid, P. Safavi-Naeini, A. Cheng. Implantable Cardioverter-Defibrillator Schocks During COVID-19 Outbreak., Journal of the American Heart Association, 2021. View Article
[3] A. Pina, S. Castellini. COVID-19 and Cardiovascular Disease: a Global Perspective., Curr Cardiol Rep, 2021. View Article
[4] Food and Drug Administration. (2023). FDA Adverse Events Reporting System (FAERS) Public Dashboard [Online]. Available: View Article
[5] Centers for Medicare & Medicaid Services. (2023) Medicare Part D Prescribers - by Geography and Drug [Online]. Available: View Article

