Volume 4 - Year 2017 - Pages 23 - 33
DOI: 10.11159/jbeb.2017.003
A Systematic Review Based Analysis of Eye-Hand Movement Onset Asynchrony Adjustments
Alexander R. Payne1, Beryl Plimmer2, T. Claire Davies3
1 Department of Mechanical Engineering, University of Auckland
Private Bag 92019, New Zealand 1142
Apay876@aucklanduni.ac.nz
2 Department of Computer Science, University of Auckland
Private Bag 92019, New Zealand 1142
B.plimmer@auckland.ac.nz
3 Department of Mechanical and Materials Engineering, Queen’s University
130 Stuart Street, Kingston, Ontario, Canada K7L 3N6
Claire.davies@queensu.ca
Abstract - When goal-directed hand movements are made, there is a common coordination pattern whereby the eyes start to move towards the target just before the hand. Onset asynchrony is the measurement of this latency. Onset asynchrony can be used to investigate the effects of neurological conditions on movement planning abilities. However, it is difficult to design a robust experiment since there is no clear understanding of why onset asynchronies change between conditions, and which elements of a task are likely to result in a change. This review aims to develop a coherent analysis of why people adjust their onset asynchrony for different situations. A systematic review methodology was employed to obtain all available research containing measurements of onset asynchrony. Searches returned 3703 articles, of which there were 38 articles that used onset asynchrony measurements as a dependent variable. In total, 30 potential onset asynchrony factors were investigated by the articles, with 25 of them affecting the timing between hand and eye onset. Generally, the eye guides the end of the previous movement, the start of the current movement, and the end of the current movement. It appears that onset asynchrony varies based on these competing requirements. When designing an experiment, one should be aware that the independent variable is likely to interact with these requirements. The experimental design must ensure that confounding factors/covariant observations are minimised.
Keywords: Eye movements, hand movements, eye-hand coordination, sensory & motor testing, onset asynchrony.
© Copyright 2017 Authors This is an Open Access article published under the Creative Commons Attribution License terms. Unrestricted use, distribution, and reproduction in any medium are permitted, provided the original work is properly cited.
Date Received: 2016-11-22
Date Accepted: 2017-02-24
Date Published: 2017-09-13
1. Introduction
Movement planning is essential for determining how to achieve a task goal for hand movements [1]. Time delays in the sensory system ensure that forward planning is required for effective control [2]. Understanding the effects of a neurological condition on movement planning ability can inform therapies [3] and design of assistive technology. However, movement planning ability is difficult to measure.
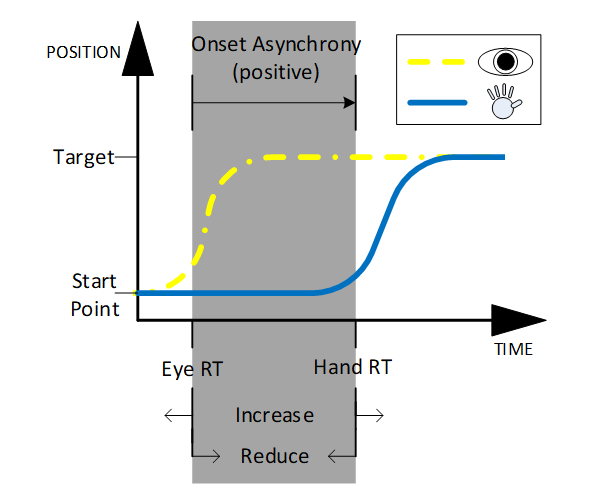
Eye-hand movement onset asynchrony (onset asynchrony for short) is one measure that relates well to movement planning ability. Onset asynchrony is a measurement of the latency between when the eyes start to saccade towards a target and when the hand movement is initiated (see Figure 1). This measurement is made by comparing two other measurements, such as hand reaction time and eye reaction time (Eq. 1).
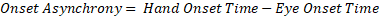
Onset asynchrony is useful for assessing typical planning behaviour since it has been shown to vary in response to different planning conditions. To investigate neurological conditions, adaptations in behaviour can be compared between participant groups. For example, Wilmut & Wann [4] investigated the ability of children with developmental coordination disorder (DCD) to integrate spatial information and pre-plan movements. They found that participants without DCD reduced their onset asynchronies when spatial information was provided pre-trial. Since this adaptation did not occur to the same extent in participants with DCD, they concluded that DCD resulted in a reduced ability to use predictive information.
However, a general understanding of which other factors can cause onset asynchronies to change, and why, is not present in the literature. This lack of understanding is a problem since onset asynchrony is not a performance measure. Unlike a measure such as task completion time, higher or lower values are not necessarily any better or worse. Therefore context is required to interpret results, but this context is currently fragmented. Onset asynchrony is interpreted in different ways and used in diverse fields such as coordination, psychology, neurology, and developmental studies.
Although many studies have examined one or two factors affecting onset asynchrony, onset asynchrony is generally used to interpret other concepts, and not to investigate the measure itself. The large number of potential factors affecting onset asynchrony precludes developing one study to determine overall factors. The contribution of this review is bringing together factors from studies in a variety of fields that would not usually be compared. We discuss these factors in relation to three functions of the gaze direction: guiding the end of a previous movement, guiding the start of the current movement, and guiding the end of the current movement.
1. 1. Conventions Adopted in This Review
Certain conventions were adopted since there was little consistency within the literature. In this review, onset asynchrony will be expressed as a one-dimensional time measurement (as opposed to expressing it as a percentage of movement time), and taken as positive when eye movement commences prior to hand movement (as per Eq. 1 and Figure 1). This eliminates the heterogeneity among articles which also describe the same measure with many names such as: movement onset asynchrony [5], onset latency [6], and difference between hand and gaze [7].
Other conventions are as follows. The hand movement for which onset asynchrony is being measured is called the current movement. The hand movement that terminated at the start point of the current movement is called the previous movement.
Movement types are classified as discrete, reciprocal, or semi-reciprocal. Discrete movements are from a start position to a target position (i.e. no previous movement). Reciprocal movements are back and forth between two targets (i.e. the previous movement was the opposite of the current movement). Semi-reciprocal movements are to a new target following on from a previous movement.
2. Systematic Review Methodology
2. 1. Definition of Comparison and Intervention
The American Academy for Cerebral Palsy and Developmental Medicine’s methodology was used as a guideline in developing this review [8]. Since the effects of the experiment set-up were being investigated, the intervention definition was quite broad. The experimental task had to involve a goal-directed hand action and saccade. The definition of the comparison was more specific; onset asynchrony.
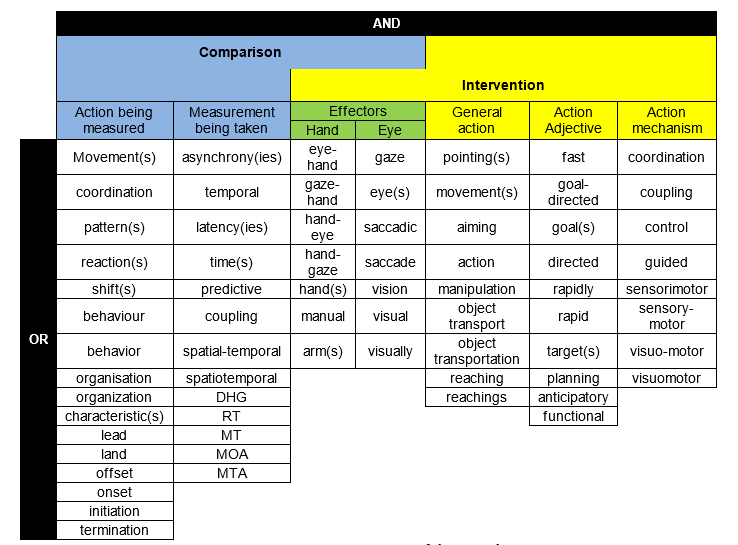
To develop a set of search terms that allowed for the broad nature of the intervention, and the multiplicity of nomenclature regarding the comparison, the search term was based on the terminology of 12 studies [5-7, 9-17]. These words were grouped together by meaning (Figure 2). Each article had to include at least one word from each column of Figure 2 in the abstract. The search also included articles mentioning eye-hand offset asynchrony, and eye and hand reaction times in their abstract, as the full article potentially included onset asynchrony too.
2. 2. Search Strategy
Once the search terms were chosen, a table of titles and rankings of results was composed. The four databases selected were EMBASE + EMBASE Classic, PubMed, Biological Abstracts, and Compendex (15-Jan-2014 was set as a cut-off date).
Inclusion criteria when reviewing abstracts were:
- - The research was peer reviewed.
- - The abstract was in English.
- - There was an allusion to a goal-directed hand action and saccade.
- - There was an allusion to temporal and spatial measurements of the hand and eye.
- - The participants were humans.
Articles were also excluded if the end effector was not displacement controlled (e.g. a joystick that controls the velocity of a cursor), or if onset asynchrony was not used as a dependent variable.
2. 3. Processing Results
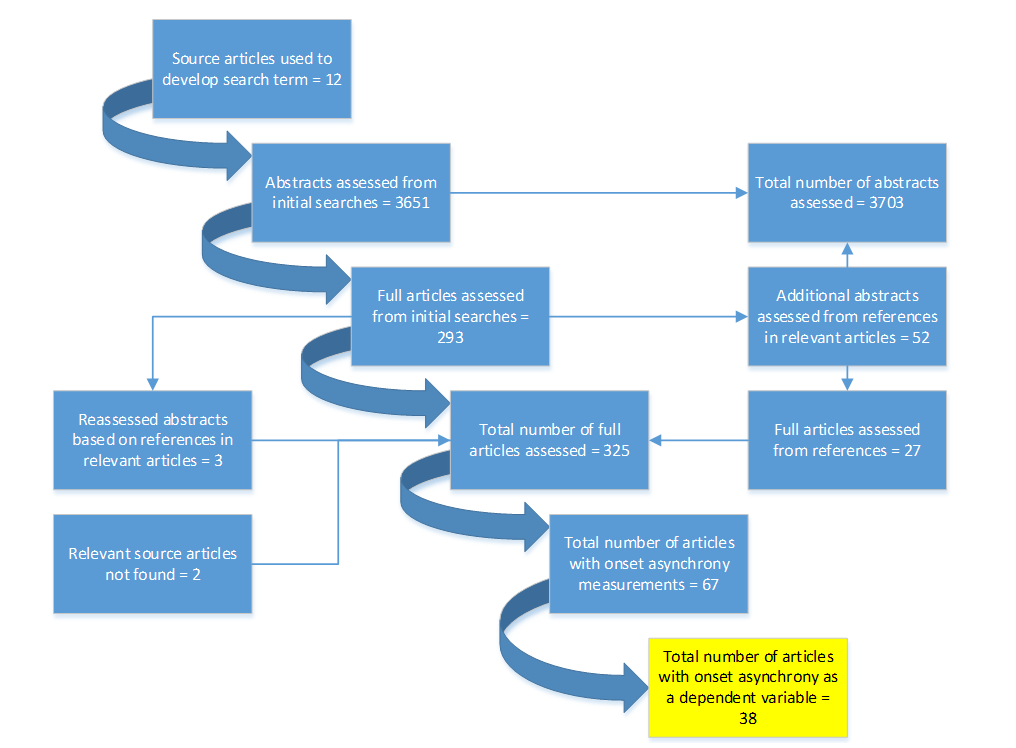
A flow chart of how the results were processed is shown in Figure 3. After full article assessment had taken place, 38 articles were identified that used onset asynchrony measurements as a dependent variable. Another 29 included onset asynchrony measurements but made no comparisons.
2. 4. Review Limitations
First, this search was evidently not exhaustive since two of the search term source articles were not found. This illustrates the limitations of searching abstracts. Second, some articles did not include a proper statistical analysis of onset asynchronies. These articles were included as they can add pieces to a complex picture. Last, little is known about the potential multi-colinearity of the onset asynchrony factors found (e.g. for reciprocal trials it is difficult to separate the effects of the current target size and the previous target size).
3. Search Results
3. 1. Results & Typical Values
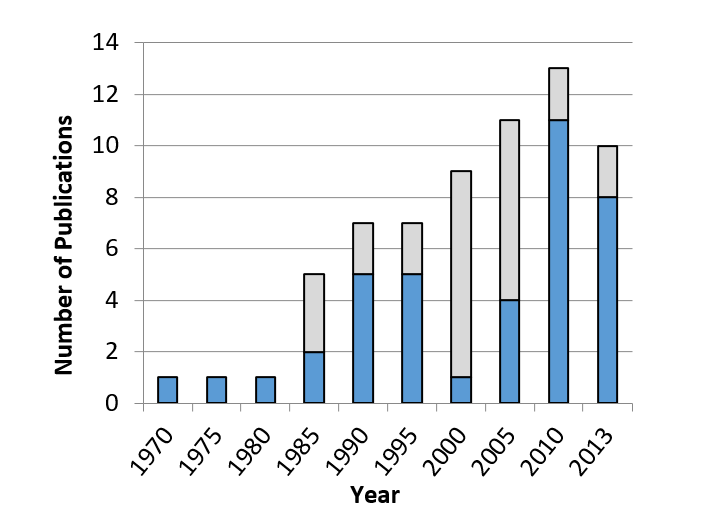
The graph in Figure 4 shows the timeliness of this review. Over the past half a century more and more published research has included measurements of onset asynchrony.
Typical values for onset asynchronies are generally positive although 17 of the total 67 articles (including the 29 articles where no comparisons were made) reported incidences of negative onset asynchronies. Magnitudes of onset asynchrony measurements are usually less than 400ms. However, in two cases [18, 19] onset asynchronies of around 1,000ms have been reported during natural continuous tasks (making tea and making a sandwich) where there were multiple points of interest. Neither speed nor accuracy was imperative for these tasks and the eyes were involved in planning a series of movements.
In experiment protocols, sampling frequencies varied from 30 – 1000Hz, and the number of participants varied from 2 to 46. Helsen et al. [20] demonstrated that a changing sampling frequency of 60Hz to 120Hz produced no difference in eye or hand onset times. Thus, it seems a sampling frequency of at least 60Hz is sufficient for accurate onset asynchrony measurements.
In general, participants’ vision was self-reported and related to foveated acuity rather than peripheral vision.
The rest of this review only concerns the 38 articles where onset asynchrony was a dependant variable. The independent variables of those articles were amalgamated into 30 different groups of onset asynchrony factors (Table 1). The main interest is whether these variables resulted in increases or decreases in onset asynchrony. This indicates a change in behaviour, whereas absolute values are an approximation of the behaviour itself.
3. 2. Factors in the Literature
In the main table of results (Table 2), factors are grouped based on how they related to task set-up: the previous target, the effector (e.g. the hand or a cursor) control, the current target, the temporal predictability, and the person making the movement. The table describes the data that exists for different factors and where there is consensus. The second column describes the condition that results in an increase in onset asynchrony (i.e. the eyes moving earlier and using hand onset as a reference point), and the third column describes the opposite. The columns to the right describe the number of studies that tend to agree or disagree with the notion expressed by the second and third columns. Studies that found no significant effect are listed under the neutral column.
Table 1. Factors affecting onset asynchrony. Note that some articles are included several times; either because they contained multiple results, or because the results were applicable to several categories. *[17] and [21] contains several different comparisons under the same category. # These articles did not perform an analysis of variance on onset asynchrony for the factor being considered.
| Factor | Condition that Increases Onset Asynchrony | Condition that Decreases Onset Asynchrony | No. Agree | No. Neutral | No. Disagree |
| Factors Relating to Previous Target | |||||
| Discrete / Reciprocal | Discrete | Reciprocal | 6 [6, 22-25] #[21] | 3 [17, 25] #[21] | |
| Previous Movement Operation | Grasping | Pointing | 1 [26] | ||
| Previous Target Size | Increased size | Decreased size | 2 [6, 27] | ||
| Previous Target Time Pressure | No time limit | Time pressure | 1 [22] | 1 [17] | |
| Factors Relating to Effector Control | |||||
| Hand Used | None | None | 3 [17, 28] #[5] | ||
| Initial Gaze Position | Separate to hand start position, closer to target location | Same as hand start position. | 1 [29] | ||
| Practice | Initial trials / Reduced practice | Practice | 1 [30] | ||
| Viewing Transformation | None | None | 1 [30] | ||
| Vision of Hand | No direct vision | Seeing hand directly | 1 [31] | 2 [28] #[32] | 1 #[32] |
| Visuomotor Mapping | Normal mapping | Novel mapping | 2 [33, 34] | ||
| Factors Relating to Current Target | |||||
| Current Target Amplitude | Decreased amplitude | Increased amplitude | 1 #[35] | 4 [31, 36] #[37, 38] | |
| Cue Prominence (Duration & Size) | Decreased cue duration / size | Increased prominence | 1 [4] | 1 [4] | |
| Current Movement Operation | Placing | Pointing | 1 [26] | ||
| Current Target Size | Increased size | Decreased size | 1 [6] | 3 [6, 27] #[37] | |
| Direction | Up, or continuing, or forward | Down, or changing direction | 4 [25, 26, 34] #[39] | 2 [31] #[38] | |
| Müller-Lyer Amplitude Illusion | None | None | 1 [28] | ||
| Target Location Predictability | Less predictable target location | Predictable target location | 2 [4] #[21] | 1 [4] | 1 #[21] |
| Target Modality | Visual target | Auditory target | 1 #[32] | ||
| Timing of Target Visibility | Reduced visibility; not visible during trial, not fixated before trial start | Increased visibility; visible during trial, previously fixated | 3 [4] #[40, 41] | 3 *[17, 25] | 1 [22] |
| Visual Field of Cue | None | None | 1 [42] | ||
| Factors Relating to Temporal Predictability of the Task | |||||
| Inter-Stimulus Interval (ISI) b/w successive targets appearing | Reduced ISI | Increased ISI | 2 [21, 25] | 1 [43] | |
| Gap Effect | Extinguished shortly before trial start | Overlap, or extinguished at trial start, or extinguished longer before trial start | 3 [44] *#[21] | 1 #[21] | |
| Timing of Trial Start Predictability | None | None | 1 [45] | ||
| Factors Relating to Participant Characteristics | |||||
| Age | Increased age | Decreased age | 2 [27] #[46] | 4 [4, 7, 27, 36] | |
| Sex | None | None | 1 [33] | ||
| Instructions / Purpose | Accuracy valued | Speed valued | 2 #[39, 47] | 1 #[48] | |
| Neurological Condition | With neurological conditions | Without neurological conditions | 6 [4, 22, 45, 46]* #[49, 50] | 2 [22, 46] | 2 #[5, 9] |
| Participant | n/a | n/a | 5 [33] #[37, 49, 51, 52] | ||
| Preferred Background Music | No music playing | Preferred music playing | 1 #[46] | ||
4. Discussion
Based on this review, we propose that onset asynchronies change according to the varying uses of vision in enabling goal-directed hand movements. Vision is clearly not used to act upon the environment, and in this context its function is to provide information to guide the hand movement. Foveated vision provides higher resolution information than peripheral vision (studies have shown preventing foveation of the target reduces movement accuracy, e.g. [37]. Therefore, it seems likely that gaze direction is influenced by what specific information is required. The timing of eye movements is influenced by when that information is required.
In the following subsections, factors are discussed in relation to three possible regions of visual interest: the end of a previous movement, the start of the current movement, and the end of the current movement. As will be shown, the visual importance of these regions seems to vary with task set-up. Additionally, onset asynchrony is often used to compare between population groups. After discussing the three areas of visual interest, factors relating to the participant are discussed.
Consideration of these areas of visual interest is important since it can inform experiment design. Interactions with independent variables can be avoided using effective experiment pre-planning. For example, say we wanted to investigate the effects of cueing the target location. We may expect cueing to reduce onset asynchrony since foveating the target location before initiating the hand movement becomes less important. Minimisation of confounding factors would suggest a discrete task rather than a reciprocal task. Reciprocal tasks generally reduce onset asynchrony but for a different reason, they introduce the need to observe the end of a previous movement. Therefore it would be difficult to detect if cueing a target location reduces onset asynchrony for reciprocal trials since attention would be drawn to the end of the previous movement regardless of cueing condition.
4. 1. Guiding the End of a Previous Movement
When the eyes are required to help guide the end of a previous movement, onset asynchrony reduces. For example, many studies show onset asynchrony is reduced for reciprocal and semi-reciprocal movements compared to discrete movements [6, 22-25]. Likewise, decreasing previous target size significantly reduced onset asynchronies [6, 27]. Rand and Stelmach [27] demonstrated this effect without the confound of current target size; they considered a movement where the current target size was held constant and only the previous target size was varied.
When the eyes are not required to guide the end of a previous movement, onset asynchrony increases. If the previous movement operation is grasping an object, tactile feedback can be used to guide the end of the previous movement. Thus, onset asynchrony increases compared to pointing [26]. This feedback may be affected by neurological condition. When participants with hemiparetic cerebral palsy grasped an object using their affected hand, onset asynchronies were reduced (and negative) compared to positive onset asynchronies when using their less affected hand, or participants without cerebral palsy [5, 9]. This is possibly due to a reduced ability to make use of tactile feedback in place of visual information.
4. 2. Observing the Start of the Current Movement
Observing the start of the current movement seems to decrease onset asynchronies. This may relate to increased monitoring of the effector at its starting position and its initial movement. Potentially this is part of calibrating the hand location and ensuring the muscle inputs made are resulting in the expected hand/effector movement outputs. For example, altering visuomotor mapping reduces onset asynchrony in novel gravity conditions [34] and reversed cursor mapping [33].
The important factor appears to be that the mapping is new, rather than differing levels of control. For example, using a dominant or non-dominant hand does not seem to affect onset asynchrony [5, 17, 28]. Likewise, increasing and decreasing gravity both resulted in decreased onset asynchrony relative to normal gravity [34] even though they change the mapping in opposite ways. However, there is one contradiction to the trend of decreased onset asynchronies in novel conditions. Changes in viewing condition (displacing vision by 10o visual angle using prisms mounted in glasses) resulted in temporarily increased onset asynchrony [30].
If observing the start of the current movement is not possible, onset asynchrony generally increases. For example, if the hand is occluded, onset asynchrony increases [31]. If observation is not required there may be a similar effect. There was no difference in onset asynchrony whether the hand was visible or occluded for a 1-DOF (degree of freedom) movement [32]. A 1-DOF movement simplifies control of the hand, reducing the need to observe it. Similarly, if initial gaze position is away from the hand and in the area of the target, there is reduced benefit in observing the hand. This increases onset asynchrony since the eyes foveate the target as quickly as possible [29]. This change was due to both an increase in hand onset time and a decrease in eye onset time, so it appears to be a change in strategy [29].
4. 3. Guiding the End of the Current Movement
Factors relating to the current target affect onset asynchrony based on whether a hand movement can be pre-planned or not, but not based on the difficulty of the movement. If a movement can be pre-planned, onset asynchrony decreases.
Prior spatial knowledge of the target allows for reduced onset asynchrony as the primary movement can be planned before trial start, and hand onset can occur earlier and closer to the eye onset. By initiating eye and hand movements at similar times, there is potentially some benefit in using the efferent from the oculomotor signal to drive the hand’s motor command [25, 53]. Previously fixating the target (rather than viewing it in peripheral vision) prior to trial start reduces onset asynchrony, regardless of whether it is to a remembered or visible target [40, 54]. Prior knowledge of target location also allows decreased onset asynchrony, as does increased cue prominence (including its duration and size) [4]. This decrease in onset asynchrony was not as prominent for individuals with DCD. Based on this interaction, a specific target location rather than just a direction may be required to pre-plan movements (and reduce onset asynchrony). In Wilmut et al. [22] where direction was predictable but amplitude was not, no significant difference in onset asynchronies between participants with and without DCD was found. Rosenbaum [55] suggests that pre-planning does not occur by multiple plans being generated and then one being selected. He supports the idea of only known parameters being used for pre-planning, and specifying unknown parameters when they become known.
While knowledge of spatial properties is valuable, the actual values of the properties are largely irrelevant to onset asynchronies. Current target size [6, 27] and amplitude [28, 31, 36] were not significant in discrete trials. Terrier et al. [6] reported current target size was significant (p<0.01) during reciprocal trials, though in this case the previous target size also varied, and Rand and Stelmach [27] found that previous target size was significant when current target size was held constant.
4. 4. The Effect of the Participant
Onset asynchrony measurements are often used to investigate differences between populations. Variations may occur due to a persons’ ability to pre-plan their movements (e.g. DCD appears to affect this) or their ability to complete a previous movement without visual feedback (e.g. as with cerebral palsy and grasping).
Increased onset asynchronies were apparent in several neurological conditions: DCD [4], schizophrenia [45], cerebellar disease [49], and Parkinson’s disease [36, 46]. As mentioned in the introduction, participants with DCD only displayed increased onset asynchronies when target locations were predictable. As such, Wilmut & Wann [4] suggest that participants with DCD have difficulties making use of target spatial information. Increased onset asynchronies also occurred in participants with advanced Parkinson’s Disease (PD) [46, 50]. There is a chance that these results are interacting with participant age (since participants with more advanced PD were generally older than those with less advanced PD) although the trials were discrete, and no other study reported a significant effect of age for discrete trials. Interestingly, when participants performed the task listening to preferred music, Sacrey et al. [46] found that participants with advanced PD exhibited different behaviour; the difference between them and other participants was somewhat attenuated.
In contrast to the increases in onset asynchrony above, two studies [5, 9] from one research group showed that when participants with hemiparetic cerebral palsy use their affected hand, they display decreased (and negative) onset asynchrony relative to participants without cerebral palsy (or when using their less affected hand). Onset asynchrony measurements were conducted when the previous movement operation was grasping, and such behaviour may not occur for different previous movement operations that do not depend on tactile feedback.
Along with neurological conditions affecting onset asynchrony, differences have also been found between individual articipants without any neurological conditions. In one study of 20 participants [33], between participant variability was significant (p<0.001). Another four studies’ [37, 49, 51, 52] suggested results vary between individuals, although they all had smaller numbers of participants. One possible cause of variations between participants is interpretation of the task instructions. Two studies [39, 47] of discrete movements found that when participants were instructed to reach a target “as fast as possible”, onset asynchrony was reduced compared to when instructed to be “as accurate as possible.” Another possible cause of variations is participant age. Older adults displayed increased onset asynchronies compared to younger ones for semi-reciprocal pointings but not discrete ones [27]. Warabi et al. [36] found age was not significant (p>0.05) for discrete tasks.
4. 5. Main Unknowns
It should be acknowledged that it is very difficult to prove that subconscious actions, such as the timing of eye movements, are engendered by intentionality or choice. Yet, these are both prerequisites of being able to adopt a strategy. Previously, it was conceived that eye and hand movements are initiated by a common central command, but more recent research shows that onset asynchrony varies with task set-up. This suggests that choice is involved, or at least that eye and hand onset times are initiated independently (Deconinck et al. [21] support this point; see Fischer and Rogal [53] for more on the common command hypothesis) meaning there is the possibility of different coordination options.
Furthermore, it seems unlikely that the timing of eye movements relative to the hand is actively used by the brain as a control variable (i.e. to adapt to a task situation). White et al. [34] considered several temporal coordination variables and found that eye onset correlated best with the peak hand acceleration. Nevertheless, how temporal coordination is established is a slightly separate question to what temporal coordination occurs; the latter can give clues to the former.
5. Conclusion
This review provides a conceptual understanding of why onset asynchronies vary across different task conditions. This understanding can be used to minimise confounding factors in the design of controlled experiments that measure onset asynchrony, such as those investigating movement planning. The review revealed 30 potential onset asynchrony factors that were investigated by 38 articles, with 25 of the factors affecting the timing between hand and eye onset. Since eyes act as a sensory input to help control the hand (the effector acting on the environment), we propose that interpretations of onset asynchrony should largely be based on the functions of the eye. In particular, the eye guides the end of the previous movement, the start of the current movement, and the end of the current movement. Onset asynchrony varies based on these competing requirements. More specifically, if there is uncertainty about the end of a previous movement then onset asynchrony reduces. Likewise, if there is uncertainty of mapping, onset asynchrony reduces. In contrast, uncertainty of the target location increases onset asynchrony. Therefore, experiment designs should consider which visual area of interest their independent variable is interacting with to minimise confounds.
References
[1] D. M. Wolpert, "Computational approaches to motor control," Trends Cogn. Sci. (Regul. Ed.), vol. 1, pp. 209-216, 1997. View Article
[2] M. Kawato, "Internal models for motor control and trajectory planning," Curr. Opin. Neurobiol., vol. 9, pp. 718-727, 1999. View Article
[3] B. Steenbergen and A. M. Gordon, "Activity limitation in hemiplegic cerebral palsy: evidence for disorders in motor planning," Developmental Medicine & Child Neurology, vol. 48, pp. 780-783, 2006. View Article
[4] K. Wilmut and J. Wann, "The use of predictive information is impaired in the actions of children and young adults with developmental coordination disorder," Experimental Brain Research, vol. 191, pp. 403-418, 2008. View Article
[5] J. Verrel, H. Bekkering and B. Steenbergen, "Eye–hand coordination during manual object transport with the affected and less affected hand in adolescents with hemiparetic cerebral palsy," Experimental Brain Research, vol. 187, pp. 107-116, 2008. View Article
[6] R. Terrier, N. Forestier, F. Berrigan, M. Germain-Robitaille, M. Lavalliere and N. Teasdale, "Effect of terminal accuracy requirements on temporal gaze-hand coordination during fast discrete and reciprocal pointings," Journal of Neuroengineering and Rehabilitation, vol. 8, pp. 10, 2011. View Article
[7] K. Rosander and C. von Hofsten, "Predictive gaze shifts elicited during observed and performed actions in 10-month-old infants and adults," Neuropsychologia, vol. 49, pp. 2911-2917, 2011. View Article
[8] J. Darrah, R. Hickman, M. O’Donnell, L. Vogtle and L. Wiart, "AACPDM methodology to develop systematic reviews of treatment interventions (Revision 1.2)," Milwaukee, WI, USA: American Academy for Cerebral Palsy and Developmental Medicine, 2008. View Article
[9] B. Steenbergen, J. Verrel and A. M. Gordon, "Motor planning in congenital hemiplegia," Disability & Rehabilitation, vol. 29, pp. 13-23, 2007. View Article
[10] G. Binsted, R. Chua, W. Helsen and D. Elliott, "Eye-hand coordination in goal-directed aiming," Human Movement Science, vol. 20, pp. 563-585, 2001. View Article
[11] S. Saavedra, A. Joshi, M. Woollacott and P. Van Donkelaar, "Eye hand coordination in children with cerebral palsy," Experimental Brain Research, vol. 192, pp. 155-165, 2009. View Article
[12] A. Ma-Wyatt, M. Stritzke and J. Trommershauser, "Eye-hand coordination while pointing rapidly under risk," Experimental Brain Research, vol. 203, pp. 131-145, 2010. View Article
[13] R. S. Johansson, G. Westling, A. Backstrom and J. Randall Flanagan, "Eye-hand coordination in object manipulation," Journal of Neuroscience, vol. 21, pp. 6917-6932, 2001. View Article
[14] M. A. Goodale, D. Pélisson and C. Prablanc, "Large adjustments in visually guided reaching do not depend on vision of the hand or perception of target displacement," Nature, vol. 320, pp. 748-750, 1986. View Article
[15] S. F. W. Neggers and H. Bekkering, "Gaze anchoring to a pointing target is present during the entire pointing movement and is driven by a non-visual signal." J. Neurophysiol., vol. 86, pp. 961-970, 2001, 2001. View Article
[16] C. Prablanc and O. Martin, "Automatic control during hand reaching at undetected two-dimensional target displacements," J. Neurophysiol., vol. 67, pp. 455-469, 1992. View Article
[17] K. Wilmut, J. P. Wann and J. H. Brown, "How active gaze informs the hand in sequential pointing 32 movements," Experimental Brain Research, vol. 175, pp. 654-666, 2006. View Article
[18] M. M. Hayhoe, A. Shrivastava, R. Mruczek and J. B. Pelz, "Visual memory and motor planning in a natural task," Journal of Vision, vol. 3, pp. 49-63, 2003. View Article
[19] M. Land, N. Mennie and J. Rusted, "The roles of vision and eye movements in the control of activities of daily living," Perception, vol. 28, pp. 1311-1328, 1999. View Article
[20] W. F. Helsen, J. L. Starkes, D. Elliott and K. L. Ricker, "Sampling frequency and the study of eye-hand coordinating in aiming," Behavior Research Methods Instruments & Computers, vol. 30, pp. 617-623, 1998. View Article
[21] F. J. A. Deconinck, V. Van Polanen, G. J. P. Savelsbergh and S. J. Bennett, "The relative timing between eye and hand in rapid sequential pointing is affected by time pressure, but not by advance knowledge," Experimental Brain Research, vol. 213, pp. 99-109, 2011. View Article
[22] K. Wilmut, J. P. Wann and J. H. Brown, "Problems in the coupling of eye and hand in the sequential movements of children with Developmental Coordination Disorder," Child: Care, Health and Development, vol. 32, pp. 665-678, 2006. View Article
[23] R. W. Angel, W. Alston and H. Garland, "Functional Relations between the Manual and Oculomotor Control Systems," Exp. Neurol., vol. 27, pp. 248-257, 1970. View Article
[24] N. Abekawa, T. Inui and H. Gomi, "Eye-hand coordination in on-line visuomotor adjustments," Neuroreport, 2013. View Article
[25] G. K. Kerr and R. J. Lockwood, "Amplitude scaling compensates for serial delays in correcting eye and arm movements," J. Mot. Behav., vol. 27, pp. 349-365, 1995. View Article
[26] J. B. J. Smeets, M. M. Hayhoe and D. H. Ballard, "Goal-directed arm movements change eye-head coordination," Experimental Brain Research, vol. 109, pp. 434-440, 1996. View Article
[27] M. K. Rand and G. E. Stelmach, "Effect of aging on coordinated eye and hand movements with two-segment sequence," Motor Control, vol. 16, pp. 447-465, 2012. View Article
[28] A. Lavrysen, D. Elliott, M. J. Buekers, P. Feys and W. F. Helsen, "Eye-hand coordination asymetries in manual aiming," J. Mot. Behav., vol. 39, pp. 9-18, 2007. View Article
[29] J. J. Adam, S. Buetti and D. Kerzel, "Coordinated Flexibility: How Initial Gaze Position Modulates Eye-Hand Coordination and Reaching," Journal of Experimental Psychology. Human Perception and Performance, vol. 38, pp. 891-901, 2012.
[30] Y. Rossetti, K. Koga and T. Mano, "Prismatic displacement of vision induces transient changes in the timing of eye-hand coordination," Percept. Psychophys., vol. 54, pp. 355-364, 1993. View Article
[31] R. Herman and R. Maulucci, "Visually triggered eye-arm movements in man," Experimental Brain Research, vol. 42, pp. 392-398, 1981. View Article
[32] J. A. Mather and J. D. Fisk, "Orienting to targets by looking and pointing: Parallels and interactions in ocular and manual performance," Quarterly Journal of Experimental Psychology Section A: Human Experimental Psychology, vol. 37A, pp. 315-338, 1985. View Article
[33] D. J. Gorbet and L. E. Sergio, "The behavioural consequences of dissociating the spatial directions of eye and arm movements," Brain Res., vol. 1284, pp. 77-88, 2009. View Article
[34] O. White, P. Lefevre, A. M. Wing, R. M. Bracewell and J. Thonnard, "Active Collisions in Altered Gravity Reveal Eye-Hand Coordination Strategies," PLoS One, vol. 7, pp. e44291, 2012. View Article
[35] E. D. Megaw and W. Armstrong, "Individual and simultaneous tracking of a step input by the horizontal saccadic eye movement and manual control systems," J. Exp. Psychol., vol. 100, pp. 18, 1973.
[36] T. Warabi, H. Noda and T. Kato, "Effect of aging on sensorimotor functions of eye and hand movements," Exp. Neurol., vol. 92, pp. 686-697, 1986. View Article
[37] R. A. Abrams, D. E. Meyer and S. Kornblum, "Eye-Hand Coordination: Oculomotor Control in Rapid Aimed Limb Movements," Journal of Experimental Psychology. Human Perception and Performance, vol. 16, pp. 248-267, 1990.
[38] C. Prablanc, J. F. Echallier, E. Komilis and M. Jeannerod, "Optimal response of eye and hand motor systems in pointing at a visual target: I. Spatio-temporal characteristics of eye and hand movements and their relationships when varying the amount of visual information," Biol. Cybern., vol. 35, pp. 113-124, 1979. View Article
[39] H. Carnahan and R. G. Marteniuk, "The temporal organization of hand, eye, and head movements during reaching and pointing," J. Mot. Behav., vol. 23, pp. 109-119, 1991. View Article
[40] C. C. Gonzalez and M. R. Burke, "The brain uses efference copy information to optimise spatial memory," Experimental Brain Research, vol. 224, pp. 189-197, 2013. View Article
[41] C. Prablanc, D. Pelisson and M. A. Goodale, "Visual control of reaching movements without vision of the limb. I. Role of retinal feedback of target position in guiding the hand," Experimental Brain Research, vol. 62, pp. 293-302, 1986. View Article
[42] S. P. Tipper, L. A. Howard and M. A. Paul, "Reaching affects saccade trajectories," Experimental Brain Research, vol. 136, pp. 241-249, 2001.
[43] G. R. Barnes and J. F. Marsden, "Anticipatory control of hand and eye movements in humans during oculo-manual tracking," J. Physiol. (Lond. ), vol. 539, pp. 317-330, 2002. View Article
[44] P. Boulinguez, J. Blouin and V. Nougier, "The gap effect for eye and hand movements in double-step pointing," Experimental Brain Research, vol. 138, pp. 352-358, 2001. View Article
[45] U. Sailer, T. Eggert, M. Strassnig, M. Riedel and A. Straube, "Predictive eye and hand movements are differentially affected by schizophrenia," Eur. Arch. Psychiatry Clin. Neurosci., vol. 257, pp. 413-422, 2007. View Article
[46] L. R. Sacrey, C. A. Clark and I. Q. Whishaw, "Music attenuates excessive visual guidance of skilled reaching in advanced but not mild Parkinson's disease," PloS One, vol. 4, pp. e6841, 2009. View Article
[47] J. D. Fisk and M. A. Goodale, "The effects of instructions to subjects on the programming of visually directed reaching movements," J. Mot. Behav., vol. 21, pp. 5-19, Mar, 1989. View Article
[48] H. Carnahan and R. G. Marteniuk, "Hand, eye, and head coordination while pointing to perturbed targets," J. Mot. Behav., vol. 26, pp. 135-146, 1994. View Article
[49] S. H. Brown, K. R. Kessler, H. Hefter, J. D. Cooke and H. Freund, "Role of the cerebellum in visuomotor coordination. I. Delayed eye and arm initiation in patients with mild cerebellar ataxia," Experimental Brain Research, vol. 94, pp. 478-488, 1993. View Article
[50] T. Warabi, N. Yanagisawa and R. Shindo, "Changes in strategy of aiming tasks in Parkinson's disease," Brain, vol. 111, pp. 497-505, 1988. View Article
[51] P. L. Gribble, S. Everling, K. Ford and A. Mattar, "Hand-eye coordination for rapid pointing movements: Arm movement direction and distance are specified prior to saccade onset," Experimental Brain Research, vol. 145, pp. 372-382, 2002. View Article
[52] N. de Bruin, L. A. Sacrey, L. A. Brown, J. Doan and I. Q. Whishaw, "Visual guidance for hand advance but not hand withdrawal in a reach-to-eat task in adult humans: reaching is a composite movement," J. Mot. Behav., vol. 40, pp. 337-346, 2008. View Article
[53] B. Fischer and L. Rogal, "Eye-hand-coordination in man: a reaction time study," Biol. Cybern., vol. 55, pp. 253-261, 1986. View Article
[54] K. Wilmut and J. Wann, "The use of prescanning in the parameterization of sequential pointing and reaching movements," J. Mot. Behav., vol. 40, pp. 558-567, 2008. View Article
[55] D. A. Rosenbaum, "Human movement initiation: specification of arm, direction, and extent," J. Exp. Psychol.: Gen., vol. 109, pp. 444, 1980. View Article
[56] J. Yordanova, V. Kolev, J. Hohnsbein and M. Falkenstein, "Sensorimotor slowing with ageing is mediated by a functional dysregulation of motor-generation processes: evidence from high-resolution event-related potentials," Brain, vol. 127, pp. 351-362, 2004. View Article
[57] C. De Boer, J. Van der Steen, R. J. Schol and J. J. M. Pel, "Repeatability of the timing of eye-hand coordinated movements across different cognitive tasks," J. Neurosci. Methods, vol. 218, pp. 131-138, 2013. View Article

